- Reaction score
- 1,667
After more than a century of searching, chemists have finally nabbed a legendary acid.
The acid called cyanoform or tricyanomethane appears widely in textbooks as one of the strongest carbon-based acids known. Yet despite attempts to make the acid dating back to 1896, cyanoform has evaded chemists until now. Researchers report September 18 in Angewandte Chemie International Editionthat they isolated the acid by figuring out crucial experimental conditions.
The main problem was temperature, says coauthor Andreas Kornath, an inorganic chemist at Ludwig Maximilian University of Munich. Researchers previously assumed that cyanoform is stable at room temperature. “It is just not,” Kornath says. Using trial and error, he and his team found that cyanoform is stable only below –40°Celsius.
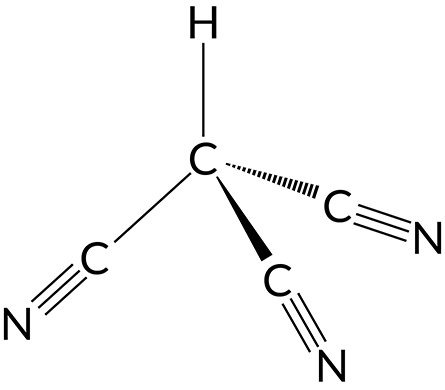
The acid has a central carbon atom attached to a hydrogen atom as well as to three cyano groups, each consisting of a carbon triple-bonded to a nitrogen. The molecule loses its hydrogen atom very easily, making it a strong acid and demonstrating a fundamental rule of carbon acids. The rule describes how electron-loving groups (in this case, the cyano groups) attached to a central hydrogen-toting carbon pull on that carbon’s electrons. The molecule’s electrons settle into a cozy position close to the cyano groups, leaving the link to the hydrogen extremely weak.
 www.sciencenews.org
www.sciencenews.org
The acid called cyanoform or tricyanomethane appears widely in textbooks as one of the strongest carbon-based acids known. Yet despite attempts to make the acid dating back to 1896, cyanoform has evaded chemists until now. Researchers report September 18 in Angewandte Chemie International Editionthat they isolated the acid by figuring out crucial experimental conditions.
The main problem was temperature, says coauthor Andreas Kornath, an inorganic chemist at Ludwig Maximilian University of Munich. Researchers previously assumed that cyanoform is stable at room temperature. “It is just not,” Kornath says. Using trial and error, he and his team found that cyanoform is stable only below –40°Celsius.
The acid has a central carbon atom attached to a hydrogen atom as well as to three cyano groups, each consisting of a carbon triple-bonded to a nitrogen. The molecule loses its hydrogen atom very easily, making it a strong acid and demonstrating a fundamental rule of carbon acids. The rule describes how electron-loving groups (in this case, the cyano groups) attached to a central hydrogen-toting carbon pull on that carbon’s electrons. The molecule’s electrons settle into a cozy position close to the cyano groups, leaving the link to the hydrogen extremely weak.

Elusive acid finally created
Cyanoform, a chemical sought for more than a century and written into textbooks, is one of the strongest organic acids.
Last edited by a moderator: